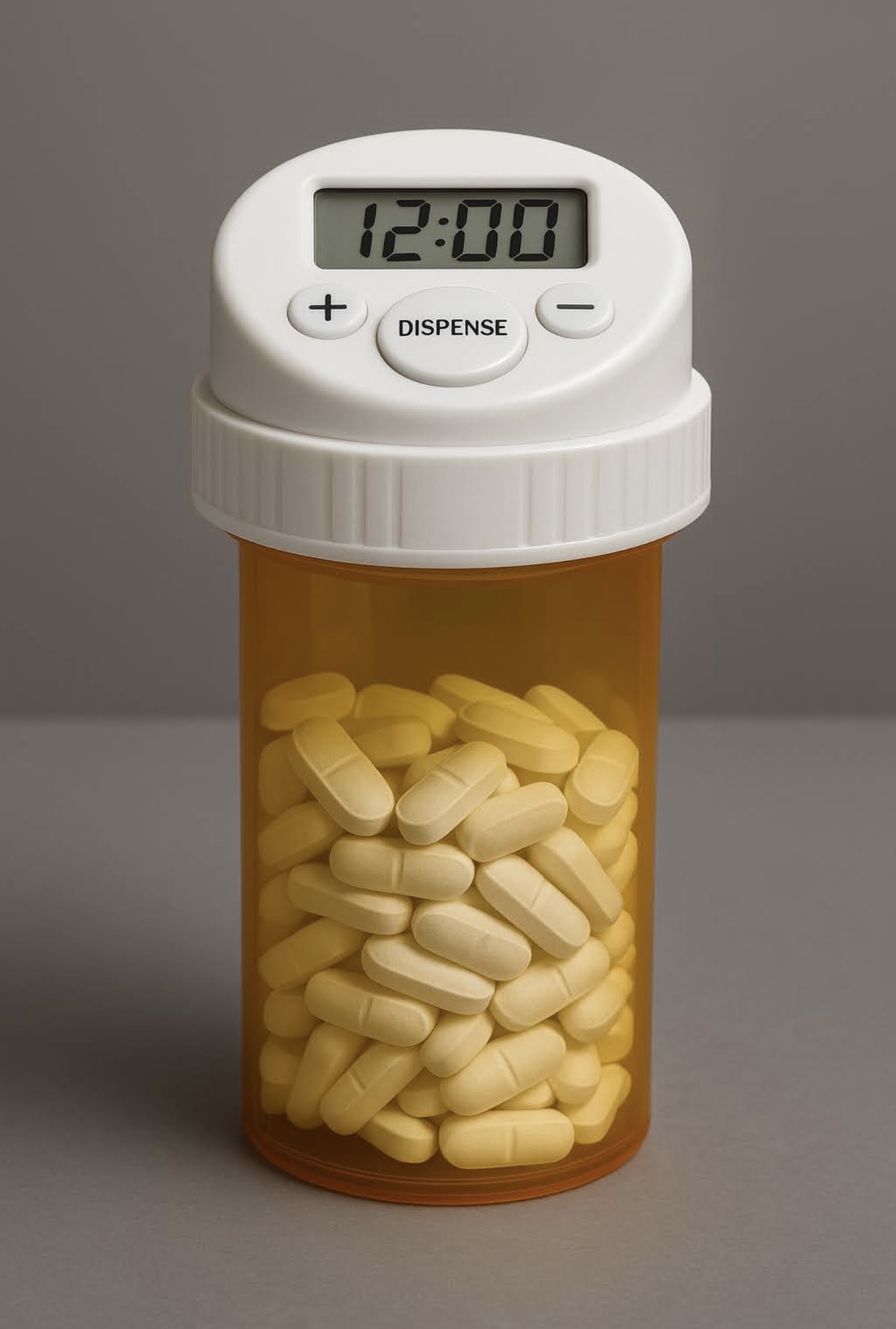
Access control, built into the cap.
FailSafe is a packaging-level, controlled-access safety cap designed to restrict physical access to prescription medications between scheduled dosing windows—helping reduce misuse, diversion, accidental pediatric exposure, and unsafe access in vulnerable settings.